Where can carbonates be found
Calcium carbonate, or CaCO3, comprises more than 4% of the earth’s crust and is found throughout the world. Its most common natural forms are chalk, limestone, and marble, produced by the sedimentation of the shells of small fossilized snails, shellfish, and coral over millions of years.
What environments do carbonate rocks form?
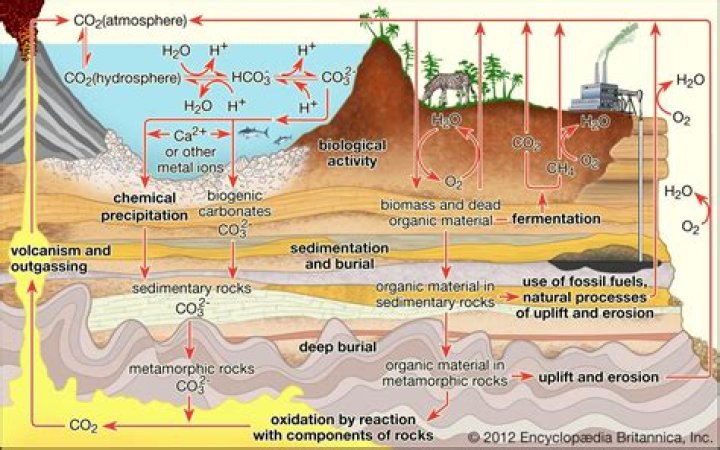
Carbonate sediments originate on land and in the sea. They are formed in three major settings: On the conti- nents, within the transitional area between land and sea, and in the shallow and deep sea. Today only araund 10 % of marine carbonate production takes place in shallow seas.
Do igneous rocks contain carbonates?
Most igneous rocks are made up of minerals which are part of the silicate group. … We have to somehow get this dissolved calcium out of the sea water and make a solid, carbonate mineral out of it so that it can sink to the seafloor and pile us as sediments to make limestone.
What is the most common carbonate?
The most common carbonate mineral in soils is calcium carbonate in the form of calcite.What are examples of carbonates?
The most common are calcite or calcium carbonate, CaCO3, the chief constituent of limestone (as well as the main component of mollusc shells and coral skeletons); dolomite, a calcium-magnesium carbonate CaMg(CO3)2; and siderite, or iron(II) carbonate, FeCO3, an important iron ore.
How are carbonate reservoirs formed?
Carbonate sediments are commonly formed in shallow, warm oceans either by direct precipitation out of seawater or by biological extraction of calcium carbonate from seawater to form skeletal material.
Which rocks are carbonates?
Carbonate rocks are also a class of sedimentary rocks that are composed primarily of carbonate minerals. The two major types of carbonate rocks are limestone (CaCO3) and dolostone, primarily composed of the mineral dolomite (CaMg(CO3)2).
How are carbonates made?
Carbonates are made from reaction between carbonic acid (aqueous carbon dioxide) and a base (or alkali). They have the formula Mx(CO3)y (e.g. Na2CO3, soda ash). Carbonate salts are generally considered weak bases, and they turn litmus paper blue.Where do carbonate sediments come from?
Carbonate sedimentary rocks are sedimentary rocks formed at (or near) the Earth’s surface by precipitation from solution at surface temperatures or by accumulation and lithification of fragments of preexisting rocks or remains of organisms.
How many carbonates are there?There are approximately 80 known carbonate minerals, but most of them are rare.
Article first time published onWhat is the most common origin of carbonate minerals in nature?
In nature, carbon atoms join with oxygen to form the carbonate ion, CO3. These ions combine with metal cations to form carbonate minerals. These minerals are commonly formed in sedimentary and oxidizing environments.
Where is dolomite used?
Uses. Dolomite is used as an ornamental stone, a concrete aggregate, and a source of magnesium oxide, as well as in the Pidgeon process for the production of magnesium.
What are carbonate reservoirs?
Carbonate reservoirs, which are typically dual or triple porosity systems, produce a major portion of the world’s oil and gas and hold more than half of the world’s largest crude oil and natural gas reserves. The best known Jurassic carbonate reservoirs are the Arab-D and Arab-C systems, Hanifa and Hadriya.
Is Quartz a carbonate?
Quartz (silicon dioxide, SiO2) is a common silicate. Carbonates have a carbon atom surrounded by three oxygen atoms. Calcite (calcium carbonate, CaCO3) is a common carbonate found in limestones.
What are carbonates composed of?
carbonate, any member of two classes of chemical compounds derived from carbonic acid or carbon dioxide (q.v.). The inorganic carbonates are salts of carbonic acid (H2CO3), containing the carbonate ion, CO2/3-, and ions of metals such as sodium or calcium.
Why is co3 called carbonate?
Carbonic Acid and Its Conjugate Bases The salt of carbonic acids are called carbonates and are characterized by the carbonate ion, CO32-. The carbonate ion is the simplest oxocarbon anion, consisting of one carbon atom surrounded by three oxygen atoms in a trigonal planar arrangement.
What is an example of metal carbonate?
Metal carbonates or hydrogen carbonates such as limestone (CaCO3), the antacid Tums (CaCO3), and baking soda (NaHCO3) are common examples. Carbonates and hydrogen carbonates decompose in the presence of acids and most decompose on heating.
What are carbonates for kids?
Quick facts for kids CarbonateSystematic name CarbonatePropertiesMolecular formulaCO2− 3Molar mass60.01 g mol-1
Where is dolomite found?
In powdered form, dolomite dissolves readily with effervescence in warm acids. Although rock beds containing dolomite are found throughout the world, the most notable quarries are located in the Midwestern United States; Ontario, Canada; Switzerland; Pamplona, Spain; and Mexico.
Where is limestone found?
Limestone-Forming Environments Most of them are found in shallow parts of the ocean between 30 degrees north latitude and 30 degrees south latitude. Limestone is forming in the Caribbean Sea, Indian Ocean, Persian Gulf, Gulf of Mexico, around Pacific Ocean islands, and within the Indonesian archipelago.
Where are evaporites found?
Typically, evaporite deposits occur in closed marine basins where evaporation exceeds inflow. The deposits often show a repeated sequence of minerals, indicating cyclic conditions with a mineralogy determined by solubility.
What is carbonate reservoir in geology?
CARBONATE. RESERVOIRS. Carbonate reservoirs account for about half of the world’s oil and gas reserves. These sedimentary reservoirs, a mixture of limestone and dolomite, include the diverse fossilized remains of the many living creatures and organisms that, in part, led to their formation.
How much of world hydrocarbons are stored in carbonate reservoirs?
Estimates show that more than 60% of the world’s oil and 40% of the its gas reserves are held in carbonate reservoirs.
What is the difference between sandstone and carbonate reservoir?
The two fundamental differences between sandstone and carbonate reservoir rocks are (1) the site of sediment production (allochthonous for sandstones vs. autochthonous for carbonates) and (2) the greater chemical reactivity of carbonate minerals (Choquette and Pray, 1970; Moore, 2001).
What is the name of co32?
Carbonate Ion is a polyatomic ion with formula of CO3(2-). Carbonate is a carbon oxoanion. It is a conjugate base of a hydrogencarbonate.
Why are carbonates bases?
Carbonates are moderately strong bases. Aqueous solutions are basic because the carbonate anion can accept a hydrogen ion from water. CO32− + H2O ⇌ HCO3− + OH− Carbonates react with acids, forming salts of the metal, gaseous carbon dioxide, and water.
Are carbonates organic?
organic compound, any of a large class of chemical compounds in which one or more atoms of carbon are covalently linked to atoms of other elements, most commonly hydrogen, oxygen, or nitrogen. The few carbon-containing compounds not classified as organic include carbides, carbonates, and cyanides.
Where do you commonly find calcite?
Description: Calcite is an abundant mineral found in many geological environments. It forms by precipitation from ground and surface waters, forming the major component of marls and limestone in the ocean, lakes and rivers.
What are metal carbonates?
Metal Carbonates are a type of base that produce a salt, water and carbon dioxide when they react with an acid. Acid + Metal Carbonates → Salt + Water + Carbon Dioxide.
What are the two most common carbonate minerals quizlet?
The two most common carbonate minerals are calcite, CaCO3 (Calcium carbonate), and dolomite, CaMg (CO3)2 (calcium/magnesium carbonate).
What is a Mineraloid give 3 examples?
A mineral-like substance that does not meet all the criteria as a true mineral. Examples include glass, coal, opal, and obsidian.