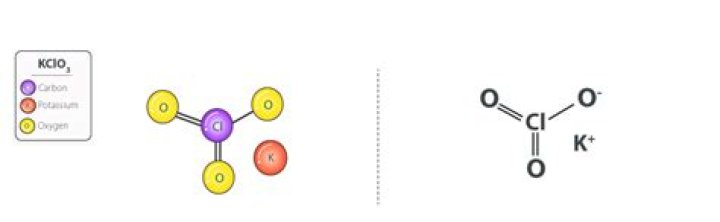
What is the name of two KClO3
Potassium chlorate | KClO3 – PubChem.
What is meant by KClO3?
Definition of potassium chlorate : a crystalline salt KClO3 that is used as an oxidizing agent in matches, fireworks, and explosives.
What is the correct name of KCl?
ChEBI Namepotassium chlorideChEBI IDCHEBI:32588DefinitionA metal chloride salt with a K(+) counterion.
What color is KCl?
The color of KCl can vary from red to white, depending on the source of the sylvinite ore. The reddish tint comes from trace amounts of iron oxide. There are no agronomic differences between the red and white forms of KCl.How do you make chlorate?
While it’s not the most efficient chemical reaction, it’s simple to make potassium chlorate by boiling bleach, cooling it, and mixing in a saturated solution of salt substitute in water. The synthesis works because potassium from the salt substitute displaces sodium from the sodium chlorate made by boiling the bleach.
What is the name for cr2o72?
Dichromate | Cr2O7-2 – PubChem.
What is potassium chlorate used for in real life?
Potassium chlorate (KClO3) is a strong oxidizing agent that has a wide variety of uses. … It is or has been a component of explosives, fireworks, safety matches, and disinfectants. As a high school or college chemistry student, you may have used it to generate oxygen in the lab.
Is chlorate the same as chloramine?
Chlorate is used in explosives and also as a pesticide. Hypochlorite and chlorine dioxide use as disinfectants are by far the principal sources in drinking water. Occurrence: … Chlorine in some form (gaseous, hypochlorite or chloramine) is used on almost 90 percent of drinking water supplies.What is the name for so42?
Sulfate is a sulfur oxoanion obtained by deprotonation of both OH groups of sulfuric acid. It has a role as a human metabolite, a Saccharomyces cerevisiae metabolite and a cofactor.
Is KCl ionic or covalent?Its chemical formula is KCl, consists of one potassium (K) atom and one chlorine (Cl) atom. An ionic compound is made of a metal element and a nonmetal element. In potassium chloride, the metal element is potassium (K) and the nonmetal element is chlorine (Cl), so we can say that KCl is an ionic compound.
Article first time published onIs KCl a chemical fertilizer?
FertilizerChemical compositionK concentration (%)KainiteKCl + NaCl + MgSO410
Is KCl a salt acid or base?
The ions from KCl derive from a strong acid (HCl) and a strong base (KOH). Therefore, neither ion will affect the acidity of the solution, so KCl is a neutral salt.
What is the name of fecl2?
ChEBI Nameiron dichlorideChEBI IDCHEBI:30812StarsThis entity has been manually annotated by the ChEBI Team.
What is flash powder made of?
A typical composition of flash powder in firework items consists of potassium perchlorate (70 wt%) as oxidizer and dark pyro aluminum (30 wt%) as fuel [6]. Other types of flash powder can also contain magnalium powder, sulfur and a diversity of oxidizers.
What's the difference between potassium chloride and potassium chlorate?
The first step is to convert sodium hypochlorite in bleach into sodium chlorate and sodium chloride. Boil a large volume of bleach (at least half a liter) until crystals start to form.
What is the name of kclo2?
PubChem CID23669246Molecular FormulaKClO2 or ClKO2SynonymsPotassium Chlorite potassium;chlorite 14314-27-3 UNII-71K32L1LFJ 71K32L1LFJ More…Molecular Weight106.55Component CompoundsCID 5462222 (Potassium) CID 24453 (Chlorous acid)
Is potassium chlorate banned?
Potassium chlorate, banned in the country since 1992, is used by some extremist organisations in making crude bombs. The chemical is sometimes used illegally to make firecrackers more noisy and colourful at only one-third the cost of legal materials.
What happens when you put a gummy bear in potassium chlorate?
Summary: When heated, potassium chlorate decomposes, producing sufficient oxygen to ignite the sugar in the gummy bear. Since the oxidation of the sugar is very exothermic, sodium chlorate continues to decompose to oxygen, and the rate of combustion becomes very rapid.
What is cr2o72 used for?
What is Dichromate? Dichromate is an anion with the chemical formula Cr2O72-. It is used as a strong oxidising agent in organic chemistry as well as a primary standard solution in volumetric analysis.
Is cr2o72 a solid?
Dichromate Cr2O72-is the conjugate base of dichromic acid formed by the removal of two protons of the acid. It is an inorganic dianion. It is a strong oxidizing agent and is interconvertible with chromate ions in solution. It is a red-orange crystalline ionic solid.
What is the correct name for ag2co3?
PubChem CID92796StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaAg2CO3 or CAg2O3SynonymsSilver carbonate Silver(I) carbonate 534-16-7 Fetizon’s reagent Disilver carbonate More…
What is SO42 in chemistry?
Sulphate (SO42-) is one of the most widely available chemical compounds accessible as naturally occurring minerals on earth. … Sulphate is basically a chemical compound that is composed of sulphur and oxygen atoms. Sulphate forms salts with a variety of elements including potassium, sodium, calcium, magnesium and barium.
What is the proper name for N2O5?
Nitrogen pentoxide | N2O5 – PubChem.
Is Lithium a chlorate?
Namesshow InChIshow SMILESPropertiesChemical formulaLiClO3
What is the difference between chlorate and perchlorate?
The key difference between chlorate and perchlorate is that chlorate is the anion derived from the dissociation of chloric acid whereas perchlorate is the anion derived from the dissociation of perchloric acid. Chlorate and perchlorate are oxyanions containing chlorine and oxygen atoms.
Is chlorate ionic or covalent?
Ammonium chlorate is an ionic compound consisting of two polyatomic ions.
Which type of bond is KCl?
Potassium chloride, KCl , is an ionic compound formed by the electrostatic force of attraction that holds the potassium cations and the chlorine anions together. Potassium, K , is located in group 1 of the Periodic Table.
Is chlorine atomic or molecular?
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature.
What is the electronegativity of KCl?
KCl: 3.0 – 0.8 = 2.2. ionic.
What is NPK fertilizer?
Every label carries three conspicuous numbers, usually right above or below the product name. These three numbers form what is called the fertilizer’s N-P-K ratio — the proportion of three plant nutrients in order: nitrogen (N), phosphorus (P) and potassium (K).
Is KCl organic or inorganic?
Potassium chloride, also known as KCl or kaon-CL 10, belongs to the class of inorganic compounds known as alkali metal chlorides. These are inorganic compounds in which the largest halogen atom is Chlorine, and the heaviest metal atom is an alkali metal.



