What does LiAlH4 do to Nitriles
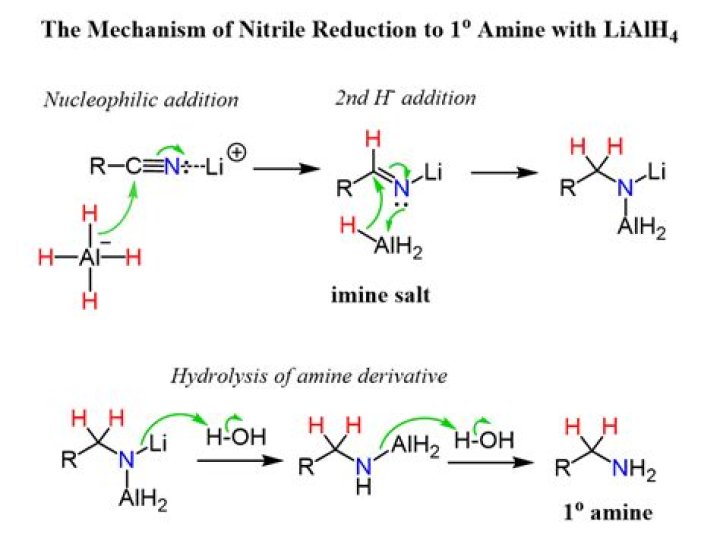
Nitriles can be converted to 1° amines by reaction with LiAlH4. During this reaction the hydride nucleophile attacks the electrophilic carbon in the nitrile to form an imine anion. Once stabilized by a Lewis acid-base complexation the imine salt can accept a second hydride to form a dianion.
Can LiAlH4 reduce a nitrile?
LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols. Amides and nitriles are reduced to amines.
How is nitrogen removed from nitrile?
After nucleophilic attack by water and a proton transfer, the nitrogen from the nitrile is removed as a leaving group in the from of ammonia (NH3). This step forms a protonated ketone called a ketonium ion which is subsequently deprotonated by ammonia to the neutral ketone and ammonium (NH4+).
What happens when you reduce a nitrile?
The overall reaction The nitrile reacts with the lithium tetrahydridoaluminate in solution in ethoxyethane (diethyl ether, or just “ether”) followed by treatment of the product of that reaction with a dilute acid. Overall, the carbon-nitrogen triple bond is reduced to give a primary amine.Can nitrile be protonated?
Nitriles can be converted to amides. This reaction can be acid or base catalyzed. In the case of acid catalysis the nitrile becomes protonated. Protonation increases the electrophilicity of the nitrile so that it will accept water, a poor nucleophile.
How do you reduce nitrile to an amine?
Nitriles can be converted to 1° amines by reaction with LiAlH4. During this reaction the hydride nucleophile attacks the electrophilic carbon in the nitrile to form an imine anion. Once stabilized by a Lewis acid-base complexation the imine salt can accept a second hydride to form a dianion.
Why is LiAlH4 a strong reducing agent?
Because aluminium is less electronegative than boron, the Al-H bond in LiAlH4 is more polar, thereby, making LiAlH4 a stronger reducing agent. Addition of a hydride anion (H:–) to an aldehyde or ketone gives an alkoxide anion, which on protonation yields the corresponding alcohol.
Can libh4 reduce nitriles?
For instance, unlike lithium aluminium hydride, lithium borohydride will reduce esters, nitriles, lactones, primary amides, and epoxides while sparing nitro groups, carbamic acids, alkyl halides, and secondary/tertiary amides.Does H2 PD reduce nitriles?
Pd/C and hydrogen will also reduce other multiple bonds, such as NO (nitro groups), CN (nitriles) and C=NR (imines).
Can lithium aluminum hydride reduce carbon nitrogen triple bond?* Lithium aluminium hydride, LAH reagent cannot reduce an isolated non-polar multiple bond like C=C. However, the double or triple bonds in conjugation with the polar multiple bonds can be reduced.
Article first time published onWhat reactions do nitriles undergo?
Reaction type: Nucleophilic Addition Nitriles typically undergo nucleophilic addition to give products that often undergo a further reaction.
Do nitriles react with amines?
Nitriles can be converted to 1° amines by reaction with LiAlH. During this reaction the hydride nucleophile attacks the electrophilic carbon in the nitrile to form an imine anion. … The dianion can then be converted to an amine by addition of water.
What happens when nitriles undergo acid hydrolysis?
When nitriles are hydrolysed you can think of them reacting with water in two stages – first to produce an amide, and then the ammonium salt of a carboxylic acid. … The nitrile is instead heated with either a dilute acid such as dilute hydrochloric acid, or with an alkali such as sodium hydroxide solution.
How do you make nitriles?
Nitriles can be made by dehydrating amides. Amides are dehydrated by heating a solid mixture of the amide and phosphorus(V) oxide, P4O10. Water is removed from the amide group to leave a nitrile group, -CN. The liquid nitrile is collected by simple distillation.
Are nitriles stable?
Aliphatic nitriles can be metabolized to the free cyanide ion, making them generally more toxic than arylnitriles which are stable in the body.
Do nitriles have resonance?
Just like carboxylic acids and carbonyl compounds, molecules called nitriles (or cyanides) also have a resonance stabilized structure and can act as both acids and bases. The carbon of nitriles bears a partial positive charge and is therefore the focus of Lewis acidity.
How is LiAlH4 a reducing agent?
Lithium aluminum hydride (LiAlH4) is a strong reducing agent. It will donate hydride (“H-”) to any C=O containing functional group. … It will reduce almost any C=O containing functional group to an alcohol.
Why is LiAlH4 a stronger reducing agent than NaBH4?
But LiAlH4 is a very strong reducing agent than NaBH4 because the Al-H bond in the LiAlH4 is weaker than the B-H bond in NaBH4. This makes the Al-H bond less stable. The reason for this is the low electronegativity of Aluminum compared to Boron. … As a result, LiAlH4 is a better hydride donor.
Does LiAlH4 reduce conjugated double?
LiAlH4 reduces a C-C double bond which is in conjugation i.e. resonance. Eg:- Styrene is reduced to ethyl benzene on reduction with LiAlH4.It also reduces C-C double bond of Cinnamaldehyde because it is in conjugation with benzene ring.
What happens when ch3ch2cn reacts with lialh4?
Answer: It leads to the formation of propanamine. Explanation: The given molecule is a propanenitrile and when it is reacted with lithium aluminium hydride , it leads to the reduction of a nitrile and forms propanamine.
Can lialh4 reduce imines?
Abstract. Atom-economical imine reduction: Classical imine-to-amine reduction uses stoichiometric quantities of LiAlH4, which generates Li/Al salt side products. The same reaction under an H2 atmosphere needs only catalytic LiAlH4 (2.5 mol %) and proceeds under surprisingly mild conditions (85 °C, 1 bar H2).
What happens when ch3cn reacts with lialh4?
The alkyl cyanide reacts with \[LiAl{H_4}\] in presence of ether followed by the treatment of product with dilute acid. Overall, we can say that the carbon-nitrogen triple bond gets reduced to give a primary amine, that is \[ – N{H_2}\] group.
Does Lindlar's catalyst reduce alkenes?
Lindlar’s catalyst is a palladium catalyst poisoned with traces of lead and quinoline, that reduce its activity such that it can only reduce alkynes, not alkenes.
What does Pd catalyst do?
Palladium on carbon is used for catalytic hydrogenations in organic synthesis. Examples include reductive amination, carbonyl reduction, nitro compound reduction, the reduction of imines and Schiff bases and debenzylation reactions.
What is Mendius reaction?
A reaction in which an organic nitrile is reduced by nascent hydrogen (e.g. from sodium in ethanol) to a primary amine:RCN+2H2 → RCH2NH2. RCN+2H2 → RCH2NH2.
How can I reduce nitriles?
The reduction of nitriles using hydrogen and a metal catalyst. The carbon-nitrogen triple bond in a nitrile can also be reduced by reaction with hydrogen gas in the presence of a variety of metal catalysts. Commonly used catalysts are palladium, platinum or nickel.
How does LiAlH4 react with esters?
Ch20: Reduction of Esters using LiAlH4 to 1o alcohols. Carboxylic esters are reduced give 2 alcohols, one from the alcohol portion of the ester and a 1o alcohol from the reduction of the carboxylate portion. Esters are less reactive towards Nu than aldehydes or ketones.
Does sodium borohydride reduce nitriles?
For example, sodium borohydride reduces nitriles in alcoholic solvents with a CoCl2 catalyst or Raney nickel. … DIBAL-H acts as a hydride source, adding a hydride ion to the carbon of the nitrile.
Does LiAlH4 change stereochemistry?
The Stereochemistry of LiAlH4 and NaBH4 Reduction The reduction of unsymmetrical ketones with LiAlH4 or NaBH4 produces a pair of stereoisomers because the hydride ion can attack either face of the planar carbonyl group: If no other chiral center are present, the product is a racemic mixture of enantiomers.
Will LiAlH4 reduce an alkene?
Lithium aluminium hydride does not reduce simple alkenes or arenes. Alkynes are reduced only if an alcohol group is nearby. It was observed that the LiAlH4 reduces the double bond in the N-allylamides.
What is lithium Aluminium hydride used for?
Uses: It is mainly used as a reducing agent in organic synthesis, and a reagent in the preparation of a variety of pharmaceuticals, organic chemicals and metal hydrides (such as NaH). It is also used as a polymerization catalyst and a propellant. It is considered as a potential hydrogen storage agent in fuel cells.



