Is hydrochloric acid a mixture
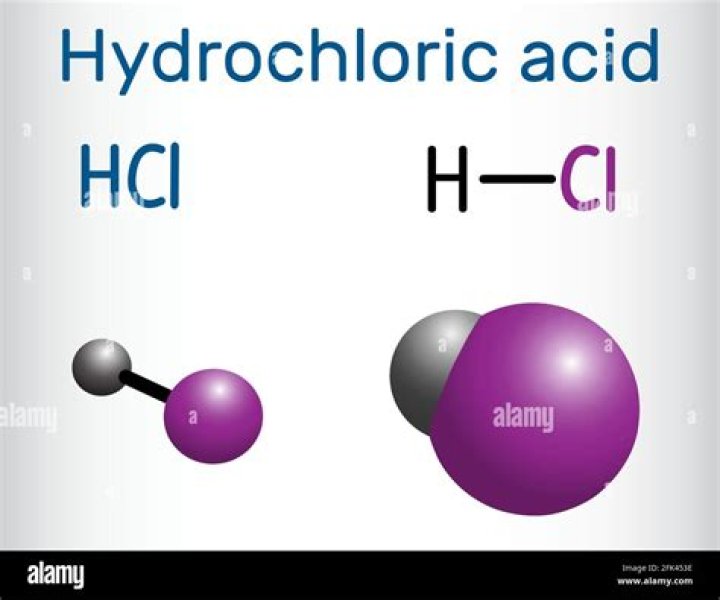
Hydrogen chloride is a mixture of hydrogen and chlorine elements. The chemical abbreviation of hydrochloric acid is HCl.
Is hydrochloric acid mixture or compound?
hydrogen chloride (HCl), a compound of the elements hydrogen and chlorine, a gas at room temperature and pressure. A solution of the gas in water is called hydrochloric acid.
Is an acid a mixture?
Aqueous acids are considered mixtures under EPCRA Sections 311 and 312. The acid itself is the hazardous chemical mixed with water when in solution.
Is hydrochloric acid a homogeneous or heterogeneous mixture?
Hydrochloric acid is a Homogeneous Mixture with the same amount of Chlorine and Hydrogen in fixed proportion throughout the solution.What type of solution is hydrochloric acid?
NamesPubChem CID313UNIIQTT17582CBUN number1789Properties
Which of these is a homogeneous mixture?
Examples of homogeneous mixtures include air, saline solution, most alloys, and bitumen. Examples of heterogeneous mixtures include sand, oil and water, and chicken noodle soup.
Is hydrochloric acid a pure substance?
Hydrochloric acid is a pure substance because it is a compound composed of two different elements, hydrogen and chlorine combined chemically in a fixed proportion. i.e., it cannot be separated by using physical methods.
Which is not a heterogeneous mixture?
A lemonade is a solution of salt (sodium chloride), sugar and lemon juice in water. It is a not a heterogeneous mixture as it has a uniform composition, as well as its components, cannot be distinguished. Whereas, others are a heterogeneous mixture.Is C4 an element compound or mixture?
As in a steam cracker, the resulting material stream is mainly made up of compounds containing four carbon atoms. But there is one substantial difference in the composition: Crude C4 contains a maximum of five percent n-butane and isobutane, while FCC-C4 contains more than 25 percent of these compounds.
Which is heterogeneous mixture?A heterogeneous mixture is a mixture in which the composition is not uniform throughout the mixture. … A heterogeneous mixture consists of two or more phases. When oil and water are combined, they do not mix evenly, but instead form two separate layers. Each of the layers is called a phase.
Article first time published onHow acids are formed?
When a hydrogen ion is released, the solution becomes acidic. When a hydroxide ion is released, the solution becomes basic. Those two special ions determine whether you are looking at an acid or a base. For example, vinegar is also called acetic acid.
How do you make an acid mixture?
Tips for Preparing Acid Solutions Always add acid to a large volume of water. The solution may then be diluted with additional water to make one liter. You’ll get an incorrect concentration if you add 1 liter of water to the acid.
Is air a mixture?
Air is nothing but a mixture of a variety of gasses. The air in the atmosphere consists of nitrogen, oxygen, which is the life-sustaining substance for animals and humans, carbon dioxide, water vapour and small amounts of other elements (argon, neon, etc.).
Why is hydrochloric acid a solution?
Hydrochloric acid (CASRN 7647-01-0) is used then released via effluent flows by the paper industry. It is a solution of hydrogen chloride (HCl) dissolved in water. HCl is a highly corrosive, strong acid, and can be a clear/colorless or light yellow liquid.
What is dilute hydrochloric acid?
Dilute hydrochloric acid is often used in the extraction of basic substances from mixtures or in the removal of basic impurities. The dilute acid converts the base such as ammonia or an organic amine into water soluble chloride salt.
Why is hydrochloric acid not a pure substance?
HCL is not a pure substance it is an acid. HCL is said to be hydrochloric acid when water is added to it as it is incomplete without water. the right answer is that hydrochloric acid is not a compound it is an acid which is full proper when we add H2O in it.
What makes a mixture a mixture?
A mixture is made when two or more substances are combined, but they are not combined chemically. There are two main categories of mixtures: homogeneous mixtures and heterogeneous mixtures.
Which of the following is a mixture?
Homogeneous mixtureHeterogeneous mixturecomposition and properties are uniform throughout the mixturecomposition and properties are non-uniform throughout the mixtureExample: Air, crude oil, ink, bloodExample: a mixture of sugar and sand, a mixture of wheat and rice
Which of these are not mixture?
A mixture is when two or more substances combine physically together. However, in water, two hydrogen atoms combine with one oxygen atom chemically, forming a new substance that has properties different from hydrogen alone or oxygen alone. … Therefore, water is not a mixture; it is a compound and it is pure.
What is homogenous and heterogenous mixture?
A mixture is composed of one or more pure substances in varying composition. … Heterogeneous mixtures have visually distinguishable components, while homogeneous mixtures appear uniform throughout. The most common type of homogenous mixture is a solution, which can be a solid, liquid, or gas.
Which one is not a homogeneous mixture?
Air is an example of a non homogeneous mixture. A non homogeneous mixture is also known as Heterogeneous Mixture. Heterogeneous mixtures can be explained as mixtures that outline no uniformity when it comes to their composition.
What is the element name of C4?
Atom NameC4Element SymbolCPDB NameC4Atom StereochemistrySIs AromaticN
How do you know if something is a mixture or a compound?
If a substance can be separated into its elements, it is a compound. If a substance is not chemically pure, it is either a heterogeneous mixture or a homogeneous mixture. If its composition is uniform throughout, it is a homogeneous mixture.
What are the 5 examples of homogeneous mixture?
- Sea water.
- Wine.
- Vinegar.
- Steel.
- Brass.
- Air.
- Natural gas.
- Blood.
What are some examples of homogeneous mixtures?
Examples of Homogeneous Mixtures include Water, Air, Steel, Detergent, Saltwater mixture, etc. Alloys are formed when two or more metals are mixed together in some specific ratio. They usually are homogeneous mixtures. Example: Brass, bronze, steel, and sterling silver.
What are 10 examples of heterogeneous mixtures?
- Cereal in milk is a great example of a heterogeneous mixture. …
- Oil and water form a heterogeneous mixture.
- Orange juice with pulp is a heterogeneous mixture. …
- Sandy water is a heterogeneous mixture. …
- A pepperoni pizza is a heterogeneous mixture.
What is difference between H * * * * * * * * * * and heterogeneous?
1. A homogenous mixture is that mixture in which the components mix with each other and its composition is uniform throughout the solution. A heterogenous mixture is that mixture in which the composition is not uniform throughout and different components are observed.
What are 5 heterogeneous mixtures?
- Concrete is a heterogeneous mixture of an aggregate: cement, and water.
- Sugar and sand form a heterogeneous mixture. …
- Ice cubes in cola form a heterogeneous mixture. …
- Salt and pepper form a heterogeneous mixture.
- Chocolate chip cookies are a heterogeneous mixture.
What kind of mixture is gasoline?
gasoline: Gasoline is a mixture of several compounds. The individual components cannot be identified from each other which means this is a homogeneous mixture.
Is HCl an acid or base?
HCl(g)Cl-(aq)Strong acidWeak base
What is acid biochemistry?
An acid is a chemical species that donates protons or hydrogen ions and/or accepts electrons. … The higher the concentration of hydrogen ions produced by an acid, the higher its acidity and the lower the pH of the solution.



