Is fluorite a face centered cubic
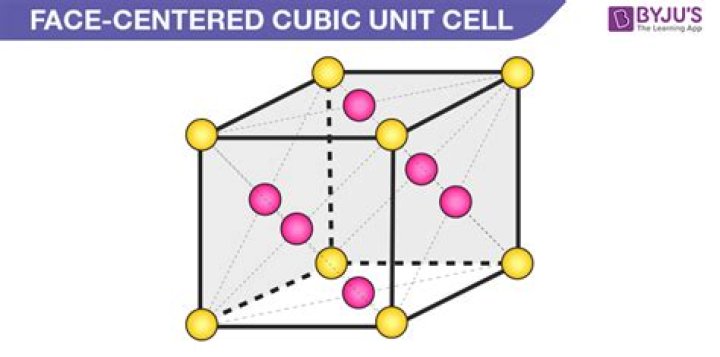
The fluorite structure, seen in calcium fluoride, has the calcium ions in a face centered cubic array with the fluoride ions in all (8) of the tetrahedral holes.
What crystal system is fluorite?
Fluorite, composition CaF2, is a halide mineral of uncommon occurrence in soils. It has a cubic crystal system with a body-centered structure. Fluorite forms perfect cubes, often with penetration twins.
Why is fluorite considered an ionic compound?
strong because of the charge difference between the two ions. Other minerals characterized by ionic bonding include fluorite, calcite and many others. … This type, electrons are shared between 2 or more ions. The classic examples illustrating this type of bonding are the molecules H2 and O2 (hydrogen gas and oxygen gas).
What type of rock is fluorite?
Fluorite forms as a late-crystallizing mineral in felsic igneous rocks typically through hydrothermal activity. It is particularly common in granitic pegmatites. It may occur as a vein deposit formed through hydrothermal activity particularly in limestones.What is cubic fluorite?
Fluorite comes in clear, blue, purple, yellow & brown and is made of cubic or octahedral crystals. It draws out negative energies and stress of all kinds. It cleanses, purifies, dispels and recognizes anything within the body that is not in perfect order. … You will receive a Fluorite crystal meaning card!
How is fluorite processed?
Just as is the case with almost ore processing and non-metal beneficiation, the concentrate fluorite is extracted by crushing, sieving, grinding, grading, flotation, filtration, drying, etc.
What is cubic fluorite structure?
The fluorite structure, which is a face-centered cubic arrangement of cations with anions occupying all the tetrahedral sites, is an open structure with interstitial voids which helps in rapid ion diffusion.
Is fluorite naturally occurring?
Fluorite is a very popular mineral, and it naturally occurs in all colors of the spectrum. It is one of the most varied colored minerals in the mineral kingdom, and the colors may be very intense and almost electric. Pure Fluorite is colorless; the color variations are caused by various impurities.Where can you find fluorite in the United States?
Fluorite is found in pegmatites from Colorado, New Mexico and Arizona. Significant fluorite from hydrothermal veins has been found in New Mexico, Colorado, Montana and Utah, although the better specimen producing states are Colorado and New Mexico.
Why is fluorite Illinois State mineral?The General Assembly made fluorite the State Mineral in 1965, when flourspar mining was a multimillion- dollar-per-year industry in Illinois. Over the years, much more fluorite has been mined in Illinois than in any other state.
Article first time published onIs fluorite igneous metamorphic or sedimentary?
Fluorite is sometimes found as a mineral in igneous rock, but it is not an igneous rock. No. Sedimentary rocks are deposited by wind, water, ice, or gravity, and they often contain fossils. Fluorite is not a sedimentary rock.
Where is fluorite sourced?
Fluorite is found worldwide in China, South Africa, Mongolia, France, Russia, and the central North America. Here, noteworthy deposits occur in Mexico, Illinois, Missouri, Kentucky and Colorado in the United States.
Is fluorite a conductor?
Fluorite is the mineral form of calcium fluoride (CaF2) and is an important host of fluorine. … We found that the electrical conductivity of fluorite is very high at moderate temperature only. The results provide direct evidence that fluorine could be an important species for electrical conduction.
Does fluorite have a definite chemical composition?
Physical Properties of FluoriteChemical ClassificationHalideDiagnostic PropertiesCleavage, hardness, specific gravity, colorChemical CompositionCaF2Crystal SystemIsometric
How many ions are in fluorite?
CaF2, Calcium Fluoride, Fluorite The unit cell has 4 Ca2+ ions and 8 F1- ions. The material is transparent in the visible spectral region, and shows electronic optical adsorption in the ultra violet and lattice optical absorption in the infrared.
What gives fluorite color?
The color of the fluorite is determined by factors including impurities, exposure to radiation, and the absence or voids of the color centers. Fluorite’s hallmark deep purple hue is the result of a small number of fluoride ions being permanently forced out of their lattice positions by irradiation or heating.
Is fluorite rare or common?
Blue fluorite is quite rare and collectors are looking for it. The brilliant yellow is very rare as well. Pink, black and colorless are the rarest fluorite colors.
Is all fluorite fluorescent?
Fluorescent Fluorite Although the name implies that all fluorites are fluorescent, this is not the case. Even specimens from the same locality will fluoresce to varying degrees or not at all. Below the octahedrons of green fluorite also fluoresce blue under LW UV light.
What is meant by fluorite structure?
In solid state chemistry, the fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic (FCC) structure.
Is rock salt a FCC?
i) Rock salt structure To summarize, the rock salt structure has ccp/fcc anions with octahedral sites fully occupied by cations and tetrahedral sites empty. … 1.26; it has six cation nearest neighbours at the six face centre positions).
What charge does fluorite have?
Property NameProperty ValueReferenceFormal Charge0Computed by PubChemComplexity2.8Computed by Cactvs 3.4.8.18 (PubChem release 2021.05.07)Isotope Atom Count0Computed by PubChemDefined Atom Stereocenter Count0Computed by PubChem
Is fluorite a renewable resource?
Fluorite reserves are limited around the world. As a non-renewable resource, fluorite is widely applied in metallurgy, chemicals and building materials.
Is fluorite the same as fluoride?
Fluorite is the crystallized version of fluorine. Fluoride is a chemical ion formed by a combination of fluorine and fluorite. In other words, fluoride is one of the components of fluorite.
What are the minerals around us?
- lead pencils (graphite)
- fertilizer (potassium, sodium, calcium)
- chalk (gypsum)
- flashbulb (zirconium)
- window glass/mirrors (silica)
- table salt (halite)
Is fluorite found in Texas?
Fluorite occurs at several other localities in Texas, notably in Hudspeth, Brewster, Presidio, Llano, and Burnet counties, but not commonly in gem quality or colors that warrant its use as gem material.
Is fluorite found in the ocean?
A Spectacular Rare Form of Fluorite The caverns formed within limestone strata that deposited in deep ocean waters millions of years ago.
Who discovered fluorite?
In the 1500s, Georgius Agricola, a German mineralogist, described a crystalline material that helped some ores melt at lower temperatures. He called it fluores, from the Latin fleure, meaning ‘to flow’. Today we know it as fluorspar, fluorite, calcium fluoride or CaF2. It was found to have a remarkable property.
Is fluorite toxic to touch?
The only chemical hazard any mineral collector needs to be aware of with fluorite is that if they are carrying out any wet chemical experimentation, the reaction between finely powdered fluorite and concentrated sulphuric can generate HF (hydrofluoric acid) which is very toxic.”
Are fluorite Octahedrons natural?
The octahedrons are undoubtedly natural crystals – not cleaved. The natural clear “rectangular” (cube) faces on fluorite are, I think, not unusual.
Does fluorite glow in the dark?
Fluorite typically glows a blue-violet color under shortwave and longwave light. Some specimens are known to glow a cream or white color. Many specimens do not fluoresce. Fluorescence in fluorite is thought to be caused by the presence of yttrium, europium, samarium [3] or organic material as activators.
What is the official state mineral?
State federal district or territoryMineralGemstoneCaliforniaGold (1965); California’s nickname is the Golden StateBenitoite (1985)ColoradoRhodochrosite (2002)Aquamarine (1971)ConnecticutAlmandine garnet (1977)DelawareSillimanite (1977)